 Patient advocates and doctors have given a warm welcome to the EU approval of a new vaccine against meningitis B, or meningococcal group B infection.
Patient advocates and doctors have given a warm welcome to the EU approval of a new vaccine against meningitis B, or meningococcal group B infection.
Vaccines already exist against the other main strains (A, C, W, and Y) that cause meningococcal disease but this is the first one widely approved for the prevention of meningitis B, a disease which mainly affects infants and young children, but can also occur in older children and young adults.
Each year, approximately 1.2 million cases of invasive meningococcal disease are recorded worldwide, of which 7,000 occur in Europe. Despite the availability of effective vaccines against several of these strains medical treatment and effective antibiotics, 8% of European patients die and some 11-19% of survivors suffer life-long consequences, including permanent brain damage, learning disabilities and hearing loss.
Routine vaccination – mainly against MenC – in many EU countries has greatly reduced the overall burden of meningococcal disease. Men B disease, while it remains relatively rare, occurs at higher incidence rates in a number of geographic regions in Europe, mainly in Belgium, Ireland, Spain and the United Kingdom.
Parents’ nightmare
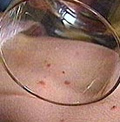
Meningitis can develop rapidly and is among many parents’ worst nightmares given the risk of death and life-long disability. Campaign groups say that existing vaccines against other strains of meningitis (such as Hib, Men C and pneumococcal) have dramatically reduced the number of cases of those diseases over the past two decades and the new Men B vaccine also has the potential to have a major impact.
Chris Head, Chief Executive of the UK Meningitis Research Foundation said: “We must not allow children to die from this disease if it can be prevented. We have a vision of a world free from meningitis and septicaemia so this news is a huge boost for our members and their families, and for doctors and scientists who have all worked tirelessly against this devastating disease.”
Professor George Griffin, professor of Infectious Diseases and Medicine at St George’s University of London said the vaccine, which is likely to be available in 2013, is a huge boost.
“This vaccine is a real step forward and could have a major impact on Men B cases. However, meningitis will remain a major clinical problem and research into microbial causes continues to be a priority alongside raising public awareness of the symptoms,” he said.
Preventable illness
For Diana Man, aged 30 and from the UK, the vaccine comes too late but she greeted the news with enthusiasm. Diana, a Paralympics reporter and herself keen sportsperson, contracted Men B in 2007 and was left without her lower legs and fingers.
“My life has changed a great deal since getting Men B, but I have learned to adjust and manage just like anyone else. If a vaccine has been produced to prevent this disease it should be made available as soon as possible to prevent anyone else having to face the difficulties myself and others face in the future both physically, emotionally and financially,” she said.
Read more:
Vaccines can spare families’ trauma
Parent’s story: take every opportunity to vaccinate your child



